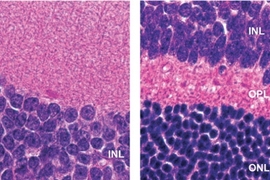
A naturally occurring DNA repair system that normally protects cells from damage can cause retinal degeneration and blindness when overstimulated, according to a new study by MIT researchers.
The research team found that relatively low-level exposure to an environmental toxic agent provoked very active DNA repair that led to surprisingly high rates of retinal degeneration in mice -- much higher than in mice lacking the same DNA repair pathway. The work raises the possibility of developing treatments for retinal degeneration by blocking a particular DNA repair pathway.
"Under some circumstances, too much DNA repair is not a good thing and could actually be a bad thing," said Leona Samson, co-director of MIT's Center for Environmental Health Sciences, professor of biology and biological engineering, and senior author of a paper on the work appearing online in the Proceedings of the National Academy of Sciences this week.
Samson and her colleagues studied a pathway involving an enzyme called Aag. The enzyme has a human analogue called AAG that may play a role in retinitis pigmentosa (RP), a group of genetic eye disorders that lead to blindness and affect between 50,000 and 100,000 people in the United States.
Aag plays a critical role in DNA repair, snipping out DNA bases damaged by environmental toxic agents, such as those found in tobacco smoke and fuel exhaust. Later on, the damaged bases are replaced with new ones.
Under normal levels of environmental toxic agent exposure, the system functions effectively. However, the researchers found that if mice were exposed to a higher (but non-lethal) level of toxic agent, the system backfired. Aag cuts out so many bases that the rest of the DNA repair system cannot keep up, leading to death of the rod and cone cells that make up the retina.
This malfunction in the DNA repair pathway occurs only in certain body tissues, including the retina, and only in mice with normal Aag levels. Mice lacking Aag do not exhibit retinal degeneration, while mice with high Aag levels had even higher levels of retinal degeneration.
Lead author of the paper is Lisiane Meira, a former research scientist in the Center for Environmental Health Sciences (CEHS). Other authors are Catherine Moroski-Erkul, technical assistant in CEHS; Stephanie Green, former graduate student in biological engineering; Jennifer Calvo, research scientist in CEHS; Dharini Shah, postdoctoral associate in CEHS; and Roderick Bronson, veterinary pathologist at Harvard Medical School.
The research was funded by the National Institutes of Health, and Samson is an American Cancer Society Professor.